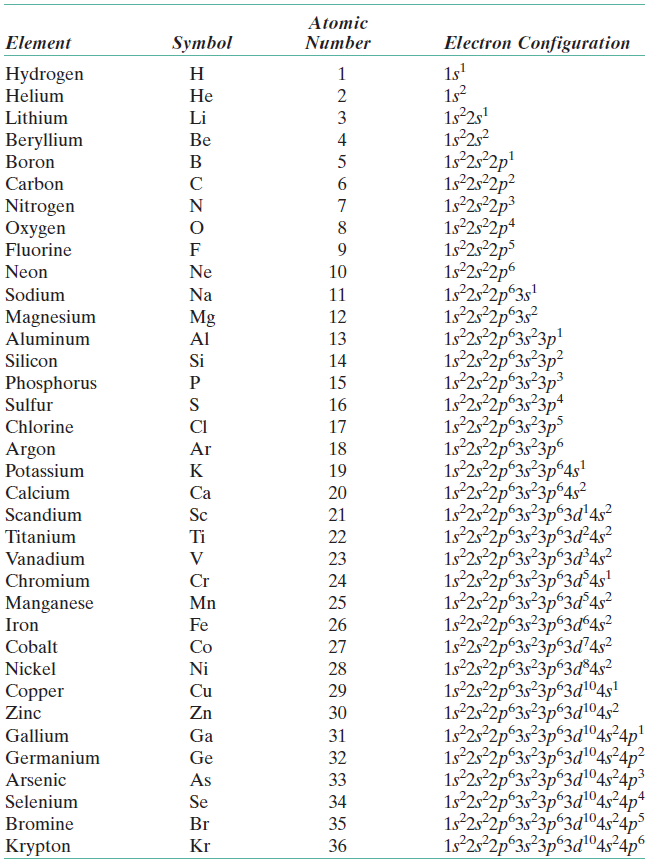
Systems with a greater number of electrons will occupy a greater amount of energy levels, meaning that they also will utilize higher energy levels. Electrons occupy orbitals that have characteristic levels of energy. Let's begin with an easy example: boron (element 5). The electron configuration is a description of where electrons are in a molecule or atom. How do we write the electron configuration of an atom? This rule is a result of electrons trying to fill the lowest energy orbitals first. Hund's Rule says that unpaired electrons must fill an unoccupied orbital before pairing up with a single electron in a previous orbital. We'll see what that looks like in the orbital diagrams below. One must spin clockwise and the other must spin counterclockwise. The Pauli Exclusion Principle states that no two electrons in the same suborbital can have the same spin. Remember that Bohr discovered that the outermost electrons in the valence shell have the greatest amount of energy, so the order of increasing sublevel energies is as follows: 1s-2s-2p-3s-3p-etc. The Aufbau Principle states that you must fill electrons in order of increasing sublevel energies. This will be super helpful when we begin writing the electron configurations from scratch, but first, there are some rules to cover for writing them. Here is a breakdown of the different subshells on the periodic table: Valence electrons occupy the outer s and p orbitals.Ĭore electrons occupy the inner d and f orbitals. Outer electrons are called valence electrons, while inner electrons are called core electrons. 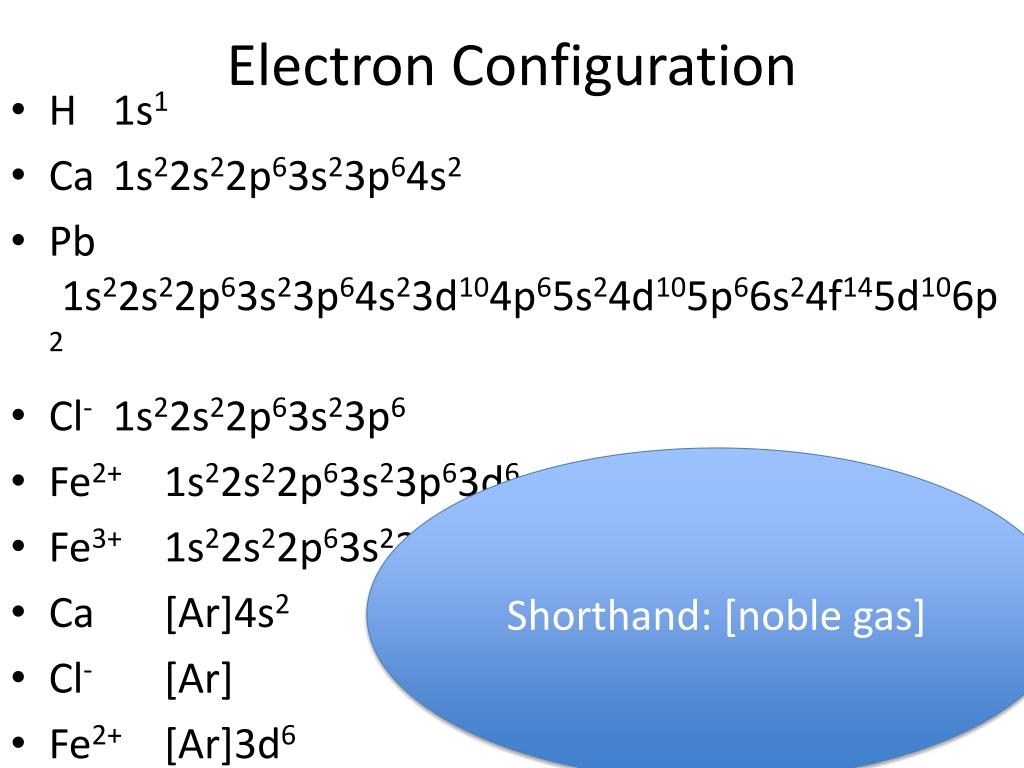
The maximum number of electrons in each subshell, respectively, are 2, 6, 10, and 14. The four different subshells are s, p, d, and f. 
Not only are the electrons in different energy levels, or shells, but they are also located in different subshells. How is the electron configuration derived? The idea behind electron configuration is quite similar to drawing out the shells in the Bohr model, in that each shell only holds a certain number of electrons. The Electron Configuration of an AtomĮlectron configuration refers to the arrangement of electrons in an atom or molecule. Taking a look at the above diagram, you can see that there is only one valence electron in the valence shell. Valence electrons are found on the valence shell of an atom, or the outermost energy level. Therefore, the valence electrons, or the outermost electrons, have the most energy. He also made the connection that the closer an electron is to the nucleus, the less energy the electron has. Each energy level corresponds to a specific energy state of the electron, which is again, fixed. This is why there are 11 electrons represented in the above diagram.īohr understood that electrons in an atom are arranged in a set of electron shells, or energy levels, around the nucleus. The atomic number of sodium is 11, which indicates that there are both 11 protons and electrons. Let's look at the Bohr model of sodium, which has 11 electrons. This causes the energy of each orbit to be fixed, quantized, or stationary. However, unlike the planets in our solar system, Bohr's orbits exist only at specific, fixed distances from the nucleus. Neils Bohr predicted that electrons orbit the nucleus in a circular orbit just like how the planets in our solar system orbit the Sun. We're back to electrons! We know that each element has a certain number of electrons, but how do we represent them? In this section, we also learn about how to properly write out the electron configuration of an element. The smaller the distance and the higher the charge, the stronger the attraction but don't worry about this yet! We'll come back to Coulomb's Law in a future unit. Magnitude of charge - The greater the charge, the stronger the attraction.ĭistance between the nuclei of the particles - The closer the two particles, the stronger the attraction. You don't have to memorize this formula, but you should understand that the strength of the forces depends on two factors: R represents the distance between the nuclei of the two particles. Q1 and q2 represent the charges of the two particles. The formula above is made up of the following variables:įe represents the calculated electric force between the two particles. The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium.Image Courtesy of APlusPhysics Breaking down Coulomb's Law The next atom is the alkali metal lithium with an atomic number of 3. The n = 1 shell is completely filled in a helium atom. The electron configuration and orbital diagram of helium are: For orbital diagrams, this means two arrows go in each box (representing two electrons in each orbital) and the arrows must point in opposite directions (representing paired spins). This is in accord with the Pauli exclusion principle: No two electrons in the same atom can have the same set of four quantum numbers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
